Blog
The Dirty Truth: Floor Contaminants in Bioprocessing and the Importance of Hose Holders
The Dirty Truth: Floor Contaminants in Bioprocessing and the Importance of Hose Holders Introduction In the world of bioprocessing, maintaining a clean and sterile environment is paramount to ensuring the safety and quality…
Read Article

The Dirty Truth: Floor Contaminants in Bioprocessing and the Importance of Hose Holders
The Dirty Truth: Floor Contaminants in Bioprocessing and the Importance of Hose Holders Introduction In the world of…
Read the Article
Press Release: New White Paper Reveals Strategies to Prevent Tubing Kinking and Ensure Fluid Transfer Efficiency
[Southampton, PA, August 24, 2023] - Kinking of tubing, a widespread challenge with far-reaching implications across industries, has been addressed…
Read the Article
Press Release: Manufacturing Day 2023
Press Release October 25, 2023- For Immediate Release Why is NewAge Industries participating in MFG Day for the first…
Read the ArticleEnhancing Thermal Management in Various Industries with Silicone Tubing
Introduction In today's fast-paced industrial landscape, efficient thermal management is essential for the optimal performance and longevity of machinery…
Read the Article
Strategies for Preventing Kinks in Tubing
Kinks in tubing can cause a lot more trouble than one would expect. They disrupt fluid flow, jeopardize product integrity,…
Read the Article
Harnessing Silicone Tubing for Effective Thermal Management in EV Batteries
Introduction As the world transitions towards a more sustainable future, electric vehicles (EVs) are at the forefront of this transformative…
Read the Article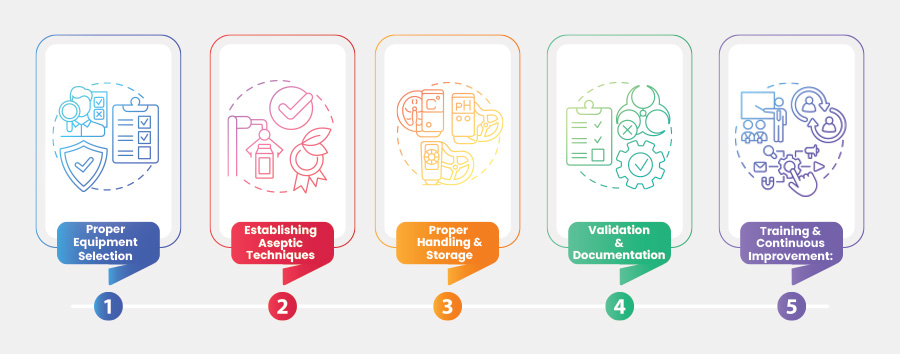
Best Practices in Fluid Transfer for Bioprocessing
Fluid transfer plays a crucial role in bioprocessing, which involves the movement of various liquids, such as media, buffers, and…
Read the Article
Biopharmaceutical Processing Basics
Demystifying the Creation Process of Biopharmaceuticals Ever wondered how life-changing treatments such as vaccines, insulin, and gene therapies are created?…
Read the Article